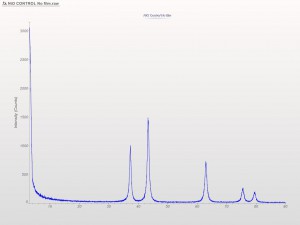
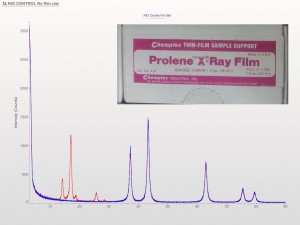
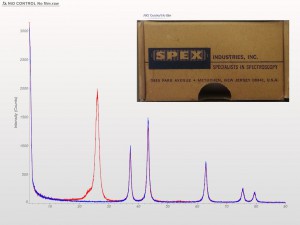
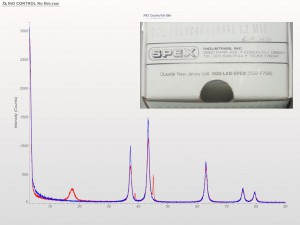
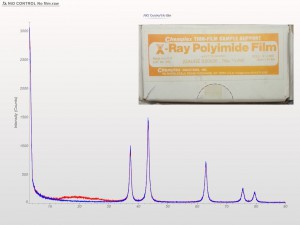
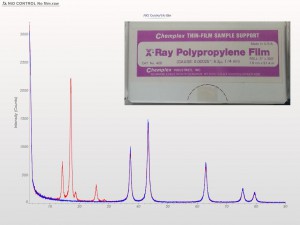
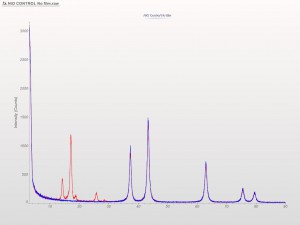
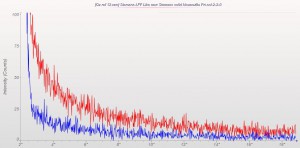
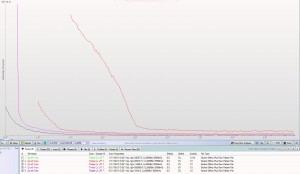
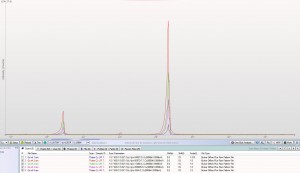
XRD patterns are complicated by a variety of undesirable effects. Some of which are easy to deal with, others are unavoidable. One of the issues we see often is scattering and diffraction effects that are actually being caused by the sample holder itself. These effects can usually be modeled out, but simply knowing which artifacts are being generated from scatter off the sample holder vs amorphous content or phases present in the sample itself can make the difference between an easy analysis and a grinding, iterative march toward a final result. One of the most common effect we see is scatter from plastic sample holders. Most of the sample holders we produce are either Aluminum or PMMA plastic, but either way, one of the easiest ways to avoid undesirable scatter is to simply enlarge the sample well. We’ve been doing this for decades on the standard, non-rotating sample holders by cutting a large, rectangular well rather than the standard, 25mm circular well.
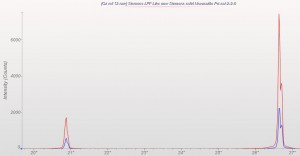
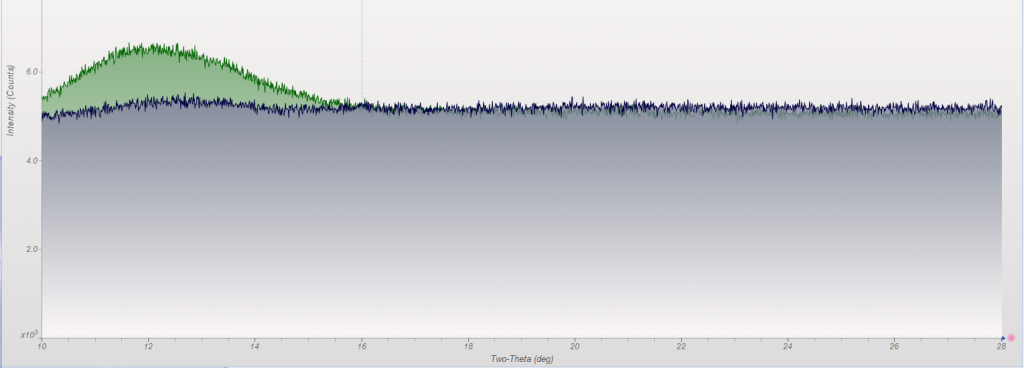
This week we did a little experiment to see just how much larger our sample well needed to be to eliminate the common PMMA hump at ~13 degrees 2Theta (Cu energy). It turns out that an increase of only 5mm in diameter made a huge difference in the total scatter even with very “wide-open” optics. See the scan images below for a real-world picture of the difference we saw. This may not seem like a significant problem until you’re looking for phases with D-spacings down in that region near the hump. Analysis of clay minerals can become particularly complicated. This is a great example of why we love talking to clients and XRD users around the world.

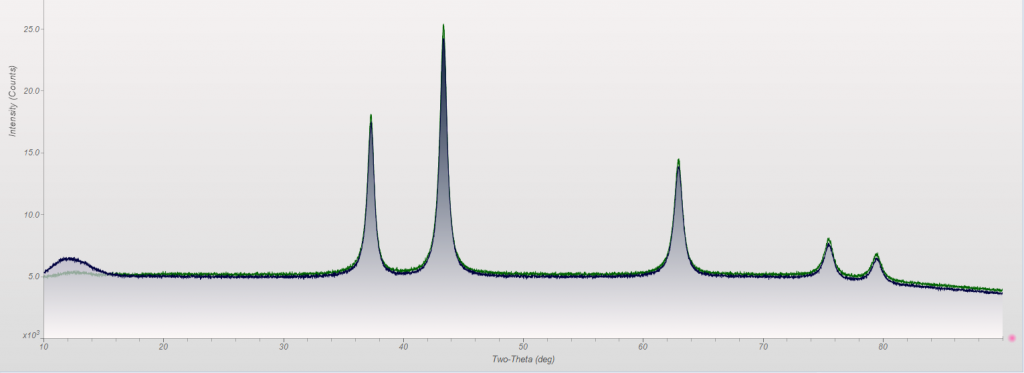
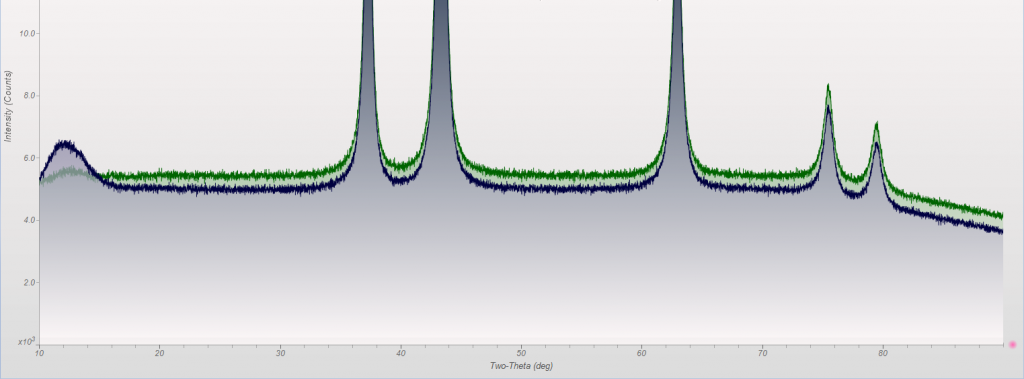




 Our recent
Our recent